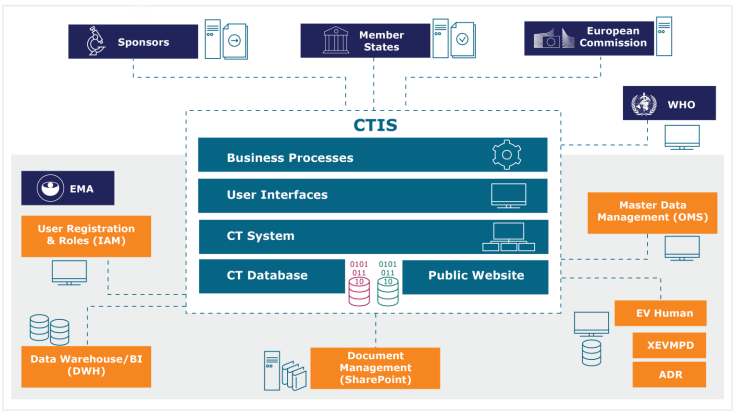
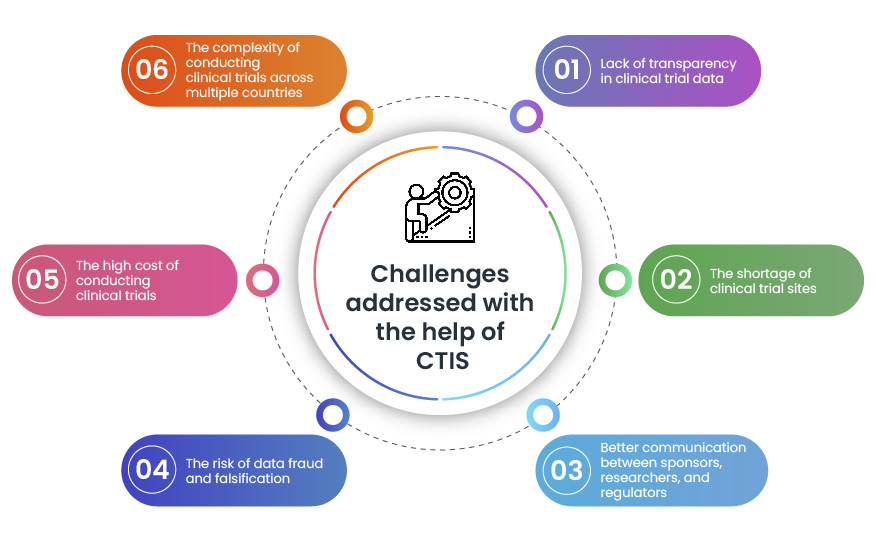
Managing the New EU Clinical Trials Regulation 536/2014 – Guidance for Navigating the Clinical Trial Information System (CTIS)

CTIS (Clinical Trials Information System). Arzt zeigt auf digitales medizinisches Interface. Text umgeben von Icons, angeordnet im Kreis. Photos | Adobe Stock

Webinaire Clinical Trials Information System (CTIS): Préparation à l'utilisation obligatoire de la Régulation des Essais Cliniques à compter du 31/01/2023

ICTD 2021 - EMA Clinical Trial Information System (CTIS) for platform trials - Noémie Manent | Ecrin