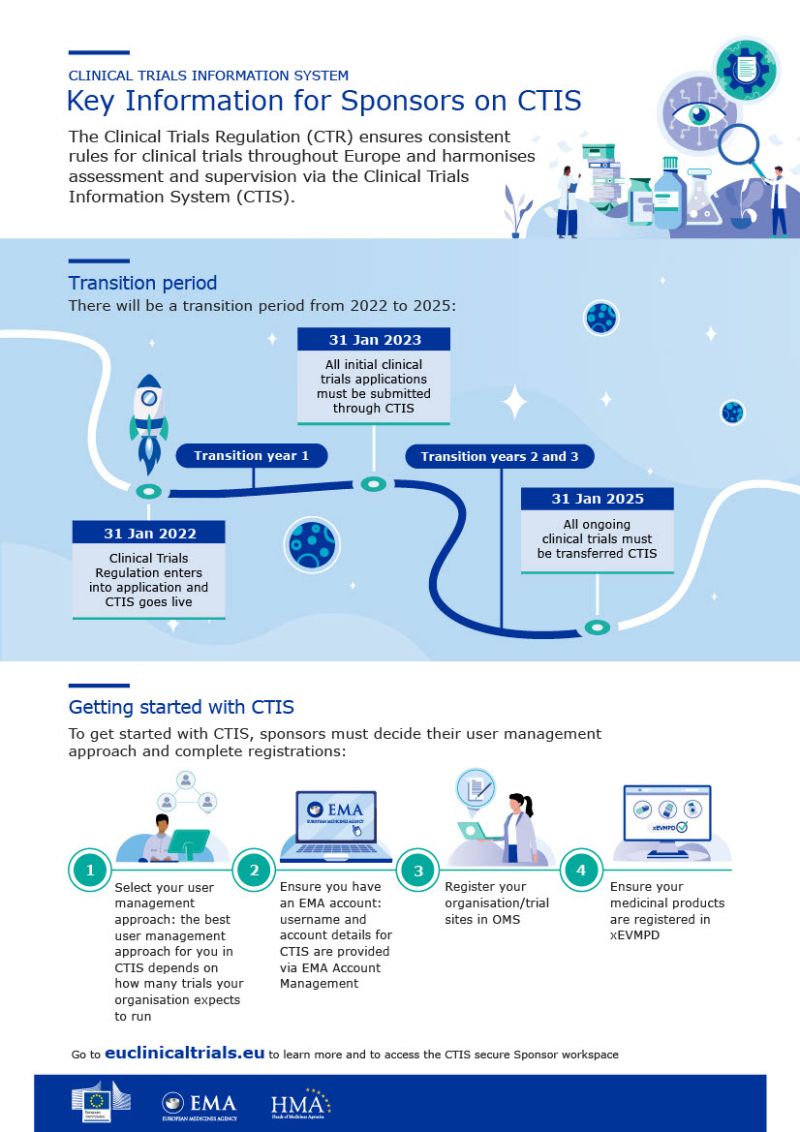
ANSM Agence nationale de sécurité du médicament et des produits de santé sur LinkedIn : Use of Clinical Trials Information System becomes mandatory for new…
.jpg)
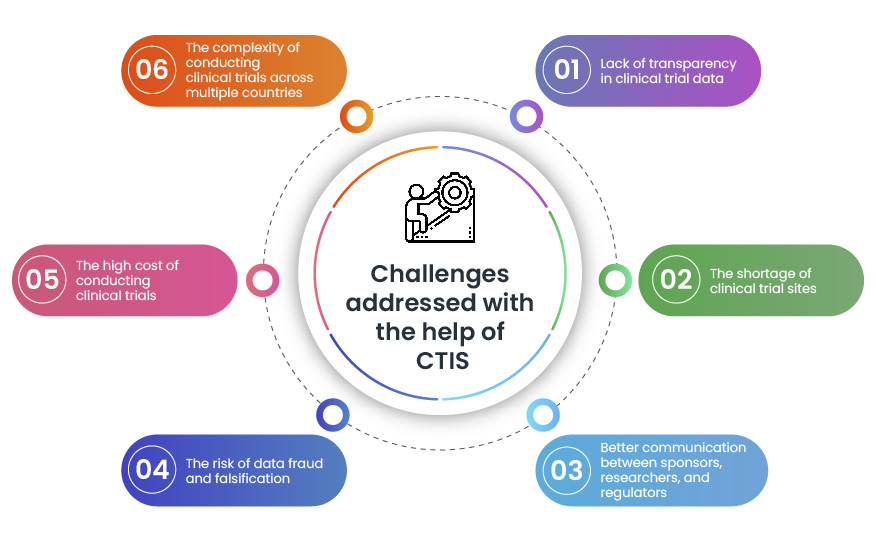
The Clinical Trial Information System (CTIS) and Data Privacy: A Guide for Clinical Trial Sponsors | Iliomad